Later years After the age of 50, Dalton performed little scientific work of distinction, although he continued to pursue research in various fields. Upper Saddle River, NJ: Pearson/Prentice Hall, 2008. Dalton’s atomic theory earned him the sobriquet father of chemistry.  
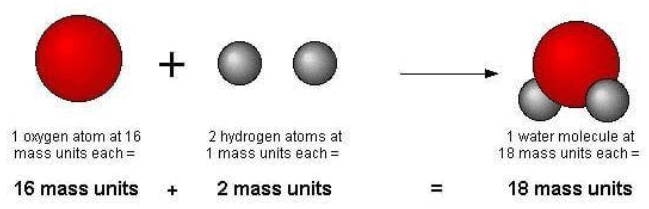
Again, this is true in chemical reactions but not necessarily in a nuclear reaction. In a chemical reaction, atoms only change the way that they are bound together with other atoms. Atoms of one element cannot change into atoms of another element. Quick Reference A theory of chemical combination, first stated by John Dalton in 1803. Atoms combine in simple, whole-number ratios to form compounds.This directly relates to Proust's Law of Definite Proportions.Ĥ. "All atomes of a given element have the same atomic number" would be a more correct statement.ģ. Daltons atomic theory also stated that all compounds were composed of combinations of these atoms in defined. Atoms of a given element are identical in size, mass, and other properties. The general tenets of this theory were as follows: All matter is composed of extremely small particles called atoms. While all atoms of an element were identical, different elements had atoms of differing size and mass. Daltons Atomic Theory is the first scientific theory to relate chemical changes to the structure, properties, and behavior of the atom. All atoms of a given element have the same mass and other properties that distinguish them from the atoms of other elements. This theory was later proved to be partially incorrect isotopes are atoms of the same element that differ in weight due to varying numbers of neutrons in their nucleus. Daltons atomic theory proposed that all matter was composed of atoms, indivisible and indestructible building blocks. The ratio of chlorine atoms in compound B compared to compound A (and. Exploration in nuclear chemistry has revealed that it is possible to destroy atoms however, atoms remain indestructible during chemical reactions.ĭalton's Atomic Theory vs. When referencing Figure 5.1d, the above can be explained by atomic theory if the copper-to-chlorine ratio in the brown compound (Figure 5.1d (b)) is 1 copper atom to 2 chlorine atoms, and the ratio in the green compound (Figure 5.1d (a)) is 1 copper atom to 1 chlorine atom. Today, we know that the even atoms are made up of smaller particles called protons, neutrons, and electrons. Each element is composed of tiny, indestructible particles called atoms. This idea dated back to the Greeks and Democritus's theories about matter. Based on a combination of their prior knowledge and his own experiments, Dalton put together the first version of his atomic theory which stated the following ideas.ġ. Dalton was mistrustful of other scientists' work and liked to assume as little as possible, but he could not have formed his atomic theory without the discoveries of Lavoisier, Priestley, and Proust.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
